The emergence of new antibiotics for drug-resistant bacteria is crucial in the ongoing battle against antibiotic resistance. As the World Health Organization has highlighted, millions of lives are at risk due to bacterial infections that have developed resistance to commonly used treatments. Innovative companies like Kinvard Bio are at the forefront of antibacterial innovation, leveraging advancements in synthetic biology to create new classes of antibiotics. These novel compounds aim to combat persistent infections that traditional antibiotics can no longer effectively treat. With a focus on addressing this public health crisis, the development of new antibiotics represents a beacon of hope in restoring efficacy against once-treatable diseases.
Introducing groundbreaking treatments for resistant infections, the field of antibacterial development has faced unprecedented challenges. In light of growing antibiotic resistance, researchers are now pivoting towards alternative strategies that utilize synthetic compounds designed to outmaneuver bacterial defenses. Kinvard Bio, for instance, embodies a fresh wave of biotechnology dedicated to pioneering innovative solutions for complex microbial threats. By refining the mechanisms of action that target bacterial ribosomes, these advancements promise to reshape the landscape of infectious disease management. The race against drug-resistant bacteria necessitates a blend of science and creativity, paving the way for a new era in effective infection control.
The Rising Threat of Antibiotic Resistance
Antibiotic resistance has evolved into one of the foremost public health challenges of our time, with its ramifications felt across the globe. According to the World Health Organization, resistant infections accounted for over a million deaths in 2019, a figure that reflects the growing concern surrounding bacterial infections that were once easily treatable. This crisis arises from the misuse and overuse of antibiotics, which encourage bacteria to develop resistance to treatments. As bacteria adapt, the once effective medications fail, resulting in higher rates of morbidity and mortality, as well as increased healthcare costs.
Healthcare professionals and researchers are increasingly alarmed by the declining efficacy of existing antibiotics. With only a handful of new antibacterial therapies entering the market each year, the medical community faces a paradox where the very tools developed to combat infections are becoming obsolete. The result is a growing number of patients with chronic and complex conditions, exacerbated by the lack of innovative alternatives. The urgent need for solutions has propelled scientific inquiry into new methods and compounds aimed at circumventing resistance mechanisms.
Innovative Solutions by Kinvard Bio
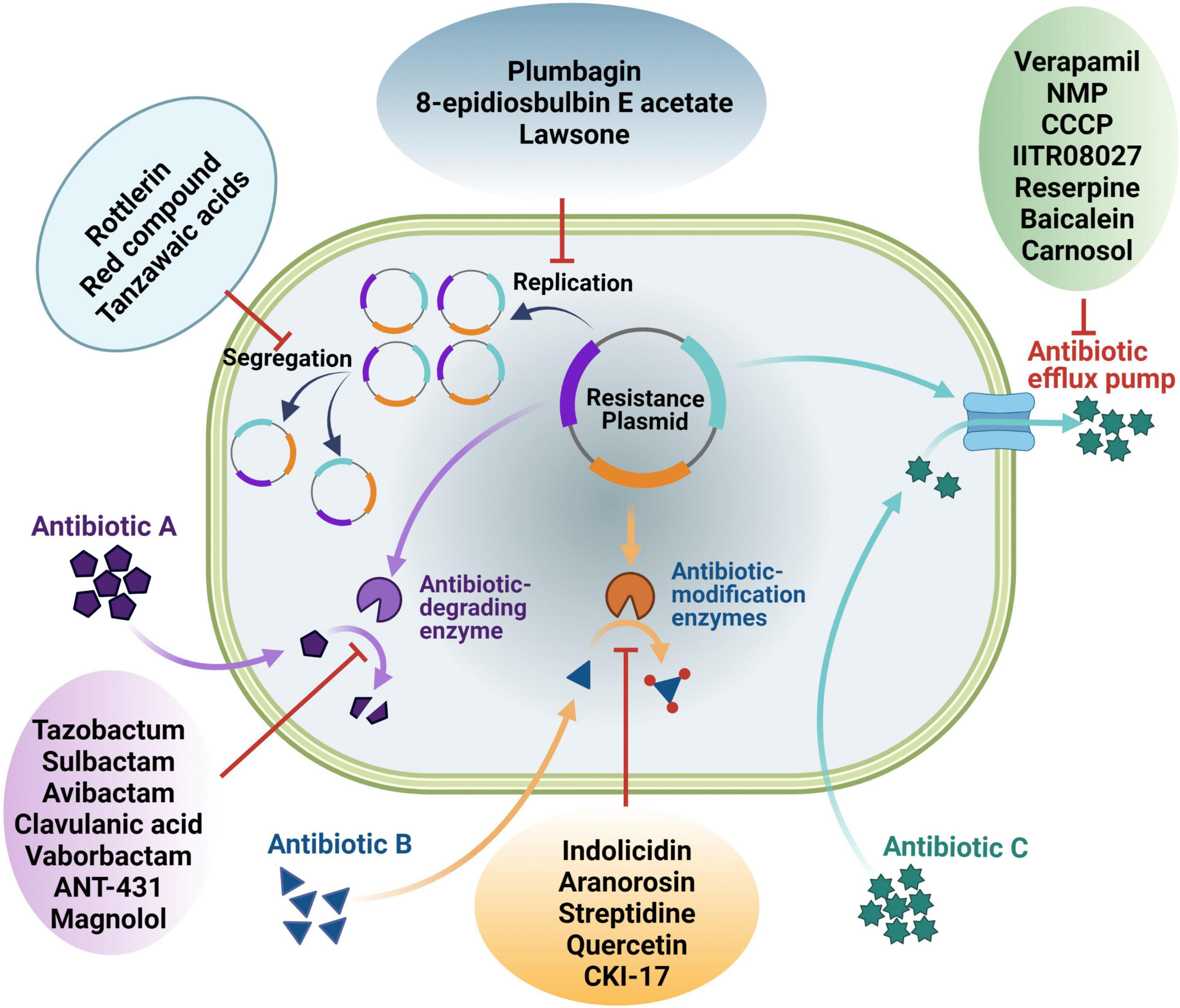
Kinvard Bio is at the forefront of the fight against antibiotic resistance, leveraging advanced synthetic biology techniques to create a new class of antibiotics. This startup, emerging from Harvard University’s Myers Lab, focuses on developing the oxepanoprolinamides, compounds that target the bacterial ribosome. This approach is significant because it opens avenues for antibiotic development that may evade mechanisms of resistance commonly found in bacteria today. By structurally optimizing these new antibiotics for effective binding, Kinvard Bio aims to restore the effectiveness of treatments against resistant strains of infections.
CEO Lloyd Payne emphasizes the importance of innovation in developing these new antibacterial agents. The compounds under development show promise not only for treating common infections but also for serious, multidrug-resistant pathogens. With funding support from various biomedical accelerators, the lab is poised to advance these antibiotics through extensive preclinical studies, moving towards clinical trials. This innovation is crucial, given the mounting evidence that traditional antibiotics are losing their battle against evolving bacterial threats.
The Role of Synthetic Biology in Antibiotic Development
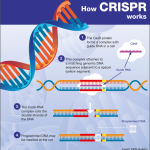
Synthetic biology has revolutionized the approach to antibiotic discovery, allowing researchers to engineer new compounds that can effectively tackle resistant bacteria. Techniques developed at institutions like Harvard foster collaborations among scientists dedicated to this goal. The work at Kinvard Bio exemplifies how synthetic biology can yield antibiotics with innovative binding properties, greatly enhancing the chances of overcoming existing bacterial resistance tactics. By utilizing advanced chemical synthesis methods, the team can create complex molecules that are structurally optimized to interact with bacterial targets.
The synthesis of such innovative antibiotics highlights the potential of combining traditional methodologies with cutting-edge technological advancements. Kinvard Bio’s efforts not only contribute to the immediate development of new treatment options but also pave the way for a greater understanding of antimicrobial resistance, potentially leading to more sustainable strategies for managing infections. This interdisciplinary approach is key to building a robust arsenal of therapies needed to combat the rising tide of antibiotic resistance.
Clinical Applications of Kinvard Bio’s Antibiotics
The clinical applications of Kinvard Bio’s new antibiotics extend beyond routine infections to tackle serious health challenges, such as complex urinary tract infections and chronic respiratory conditions. Initial preclinical trials have demonstrated their efficacy against a broad spectrum of pathogens, particularly those resistant to existing medications. This could dramatically change the landscape of treatment options available to healthcare providers, offering new hope to patients who currently have limited resources in managing their conditions.
By focusing on both intravenous and oral formulations, Kinvard Bio addresses the need for versatile treatment options. Oral antibiotics can be particularly valuable in outpatient settings, significantly reducing healthcare burdens associated with hospitalization. Given that prolonged stays in medical facilities can heighten the risk of acquiring secondary infections, these advancements could lead to better patient outcomes and fewer complications across diverse clinical scenarios.
Challenges in Approving New Antibiotics
The pathway to the approval of new antibiotics is fraught with challenges, primarily due to the slow pace of innovation in the biopharmaceutical sector. Between 2017 and 2022, only a small number of antibiotics were approved, with many falling short of being wholly new drugs. This stagnation raises concerns about the future availability of effective treatments and emphasizes the urgent need for funding and resources to support the research and development of novel compounds.
The hurdles are multifaceted, ranging from regulatory requirements to the economic viability of investing in antibiotic development. Unlike chronic disease medications, antibiotics are typically used for short durations, which conversely reduces potential profits. As a result, many pharmaceutical companies shy away from investing heavily in antibiotic research. Overcoming these barriers requires collaboration between governmental bodies, research institutions, and private companies to incentivize the development of new antibacterial innovations.
Funding and Support for Antibacterial Innovation
Financial backing is essential for the continued advancement of novel antibiotics, especially in the face of rising resistance. Initiatives like CARB-X have become critical in providing necessary resources to startups like Kinvard Bio that focus on developing new treatments for drug-resistant infections. Such funding not only enhances research capabilities but also fosters a supportive ecosystem for innovation, encouraging scientists and entrepreneurs to pursue ambitious projects aimed at tackling antibiotic resistance.
In addition to grants, partnerships with established biopharmaceutical firms and academic institutions can propel forward the mission of developing effective new antibiotics. As Kinvard Bio actively seeks collaboration with entities like the Blavatnik Biomedical Accelerator, they illustrate the importance of networking within the scientific community. By pooling expertise and resources, these partnerships can lead to breakthroughs that may otherwise be unattainable in isolation.
The Future of Antibiotic Discovery
Despite the significant challenges, the future of antibiotic discovery remains promising, driven in part by organizations like Kinvard Bio that are committed to addressing antibiotic resistance. With the potential for new antibiotic classes to emerge, scientists can develop tailored treatments that can keep pace with evolving bacterial pathogens. The ongoing commitment to research, innovation, and collaboration is crucial to reversing the trend of increasing resistance and ensuring accessible antibiotics for future generations.
As the landscape of medicine continues to evolve, the integration of synthetic biology and modern chemistry will play an indispensable role in shaping the future of antibacterial therapy. With projects underway that focus on innovative approaches to drug development, there is a glimmer of hope that the tide of antibiotic resistance can be turned, offering renewed prospects for patients suffering from resistant infections.
Collaboration in the Antimicrobial Sector
Collaboration is pivotal to the success of combating antibiotic resistance. As evidenced by Kinvard Bio’s formation in a research-rich environment at Harvard, multidisciplinary partnerships yield innovative solutions that can tackle complex health challenges. Engaging scientists, clinicians, and policymakers fosters a holistic approach to addressing the crisis surrounding drug-resistant bacteria, warranting the alignment of expertise and perspectives from various sectors.
In today’s interconnected world, it is essential for academic institutions, biotechnology companies, and health organizations to work together more than ever. The convergence of ideas can promote faster development times for new antibiotics, enhance clinical trial designs, and ultimately improve patient outcomes. By fostering a culture of collaboration, the antimicrobial sector can create a formidable front against the escalating threats posed by resistant pathogens.
Public Awareness and Antibiotic Stewardship
Raising public awareness regarding antibiotic resistance is imperative for fostering responsible use of these critical medications. Campaigns aimed at educating the general population about the importance of proper antibiotic usage and the consequences of misuse can help mitigate the emergence of resistant bacteria. By empowering patients and healthcare providers with knowledge, the cycle of over-prescription and inappropriate use can be diminished.
Antibiotic stewardship programs, implemented by healthcare facilities, aim to monitor and optimize antibiotic prescriptions. By promoting the judicious use of antibiotics, these initiatives play a crucial role in maintaining the effectiveness of existing treatments while also paving the way for new antibiotics like those being developed by Kinvard Bio. As awareness grows, the synergy between education and innovation can foster an environment for responsible antibiotic use and a sustainable response to the ongoing challenge of antimicrobial resistance.
Frequently Asked Questions
What are new antibiotics for drug-resistant bacteria being developed by Kinvard Bio?
Kinvard Bio is developing a new class of antibiotics called oxepanoprolinamides, which target the bacterial ribosome. These antibiotics are designed to bind effectively to their target in a unique manner, providing potential solutions to combat drug-resistant infections.
How does Kinvard Bio aim to fight antibiotic resistance through its new antibiotics?
Kinvard Bio aims to address antibiotic resistance by creating innovative antibacterial compounds that bind differently to the bacterial ribosome, minimizing the chances of pre-existing resistance mechanisms rendering them ineffective against drug-resistant pathogens.
What is the significance of synthetic biology in the development of new antibiotics for drug-resistant bacteria?
Synthetic biology plays a crucial role in the development of new antibiotics, such as those created by Kinvard Bio, by enabling scientists to engineer complex molecules that can effectively target and combat drug-resistant bacterial infections.
Why is there a critical need for new antibiotics for bacterial infections?
The critical need for new antibiotics arises from the increasing prevalence of antibiotic resistance, which has rendered many existing treatments ineffective and led to high mortality rates from bacterial infections.
How do Kinvard Bio’s antibiotics differ from existing antibacterial treatments?
Kinvard Bio’s antibiotics differ from existing treatments by utilizing a novel binding mechanism to the bacterial ribosome, which helps avoid resistance pathways that other antibiotics fall prey to, thereby offering a fresh approach to treating resistant infections.
What types of infections are Kinvard Bio’s new antibiotics targeting?
Kinvard Bio’s new antibiotics are targeting a range of acute and chronic infections, including bacterial pneumonia, complicated urinary tract infections, and chronic respiratory infections, particularly those associated with significant unmet medical needs.
What role does Kineticos Life Sciences play in the development of new antibiotics at Kinvard Bio?
Kineticos Life Sciences provides funding and support for Kinvard Bio, facilitating the translation of innovative research into viable new antibiotics that effectively address the challenges of antimicrobial resistance.
How successful have the early studies been for Kinvard Bio’s new class of antibiotics?
Early preclinical studies have shown that Kinvard Bio’s new antibiotics are active against a broad range of pathogens, including those that are resistant to existing antibiotics, indicating promising potential in treating serious bacterial infections.
What future applications are anticipated for Kinvard Bio’s new antibiotics?
Future applications may expand to include treatments for chronic infections such as non-tuberculous mycobacteria lung disease, potentially addressing even more difficult-to-treat infections caused by drug-resistant bacteria.
How do new antibiotics for drug-resistant bacteria affect hospital stay and patient outcomes?
New antibiotics for drug-resistant bacteria, like those developed by Kinvard Bio, can reduce hospital admissions and lengths of stay by providing effective oral and intravenous treatment options, thus lowering the risk of acquiring new infections during hospitalization.
| Key Topic | Details |
|---|---|
| Kinvard Bio and New Antibiotics | Kinvard Bio, launched from Harvard, focuses on creating new antibiotics targeting drug-resistant infections. |
| Antibiotic Resistance Crisis | Antibiotic resistance is responsible for over 1 million deaths annually, highlighting a critical need for new treatment options. |
| Current Antibiotic Development | Only a dozen antibiotics were approved globally between 2017-2022, with just two being from new classes. |
| Kinvard’s Research Approach | The research focuses on bacterial ribosome targeting, developing oxepanoprolinamides with unique binding properties. |
| Funding and Support | Kinvard Bio has received significant support from organizations like CARB-X and the Blavatnik Biomedical Accelerator. |
| Future Prospects | Kinvard aims to develop treatments for acute and chronic bacterial infections, with potential for a wide range of formulations. |
Summary
New antibiotics for drug-resistant bacteria are crucial in addressing the growing health crisis due to antibiotic resistance. Kinvard Bio is at the forefront of this effort, developing innovative antibiotic solutions targeting resistant pathogens. With profound implications for global health, these new antibiotics could restore effective treatment options for infectious diseases that have increasingly become difficult to manage with existing medications. Their research not only brings hope for tackling current resistant infections but also prepares a foundation for future breakthroughs in antibiotic therapies.